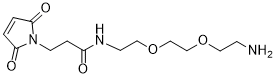
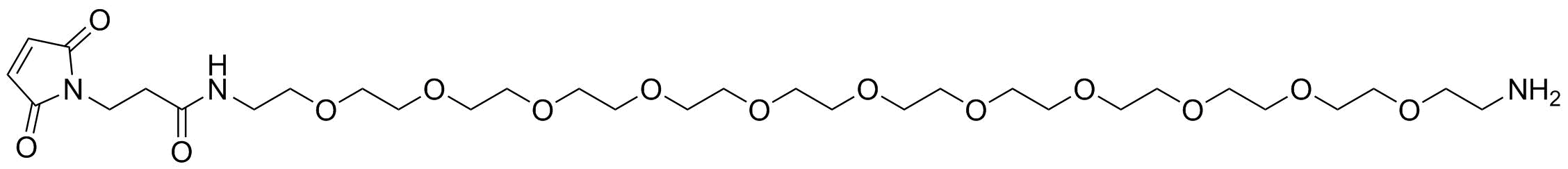
Mal-amido-PEG-amine contains two important functional groups, namely a maleimide group (-Mal) and an amine group (-NH2).
The maleimide group is known for its ability to react selectively with thiol-containing molecules, such as cysteine residues in proteins, via a Michael addition reaction. This property allows for the specific conjugation of Mal-amido-PEG-amine to thiol-containing biomolecules, resulting in the formation of stable and well-defined conjugates.
The amine group is a versatile functional group that can be easily modified or conjugated with a wide range of functional molecules, such as carboxylic acids, aldehydes, and isocyanates. This makes Mal-amido-PEG-amine a useful tool for the development of drug delivery systems and biomaterials.
Mal-amido-PEG-amine has been utilized in the development of various drug delivery systems, such as antibody-drug conjugates (ADCs) and protein-polymer conjugates. In ADCs, the amine group of Mal-amido-PEG-amine is conjugated to the antibody via an amide bond, while the maleimide group is used to selectively conjugate a cytotoxic drug to the antibody via a thiol-maleimide reaction. This results in a highly specific and effective drug delivery system that targets cancer cells while sparing normal cells.
Mal-amido-PEG-amine
| Cat# | Name | Structure | Pricing |
|---|---|---|---|
| AP10745 | Mal-amido-PEG2-amine | Pricing | |
| AP11479 | Mal-amido-PEG9-amine TFA salt | Pricing | |
| AP13273 | Mal-amido-PEG11-amine TFA salt | Pricing | |
| AP13272 | Mal-amido-PEG23-amine | Pricing |