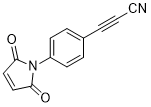
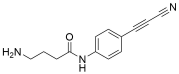
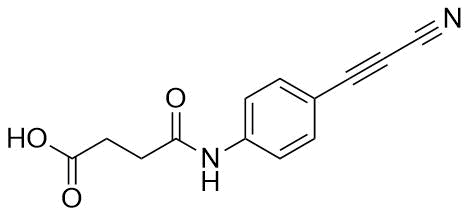
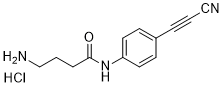
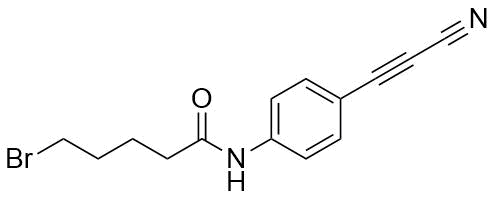
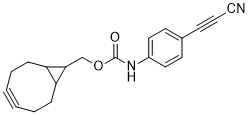
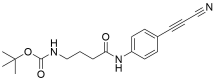
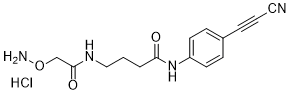
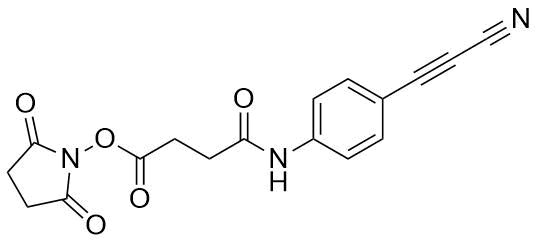
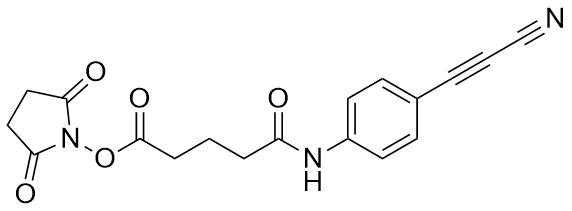
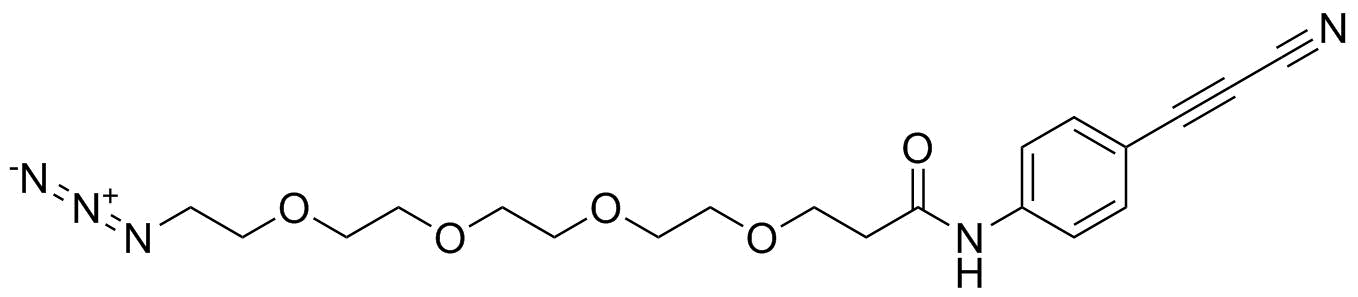
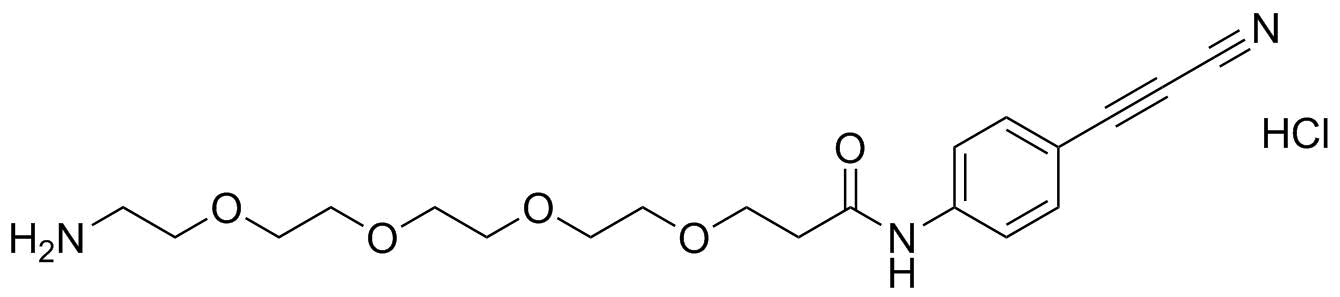
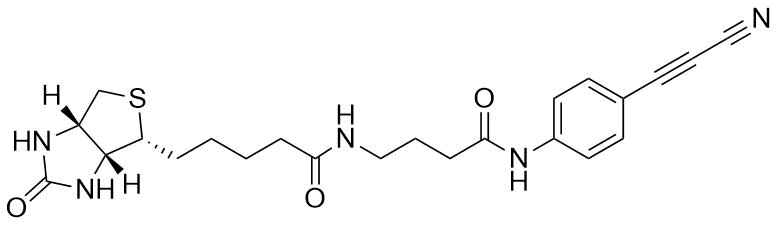
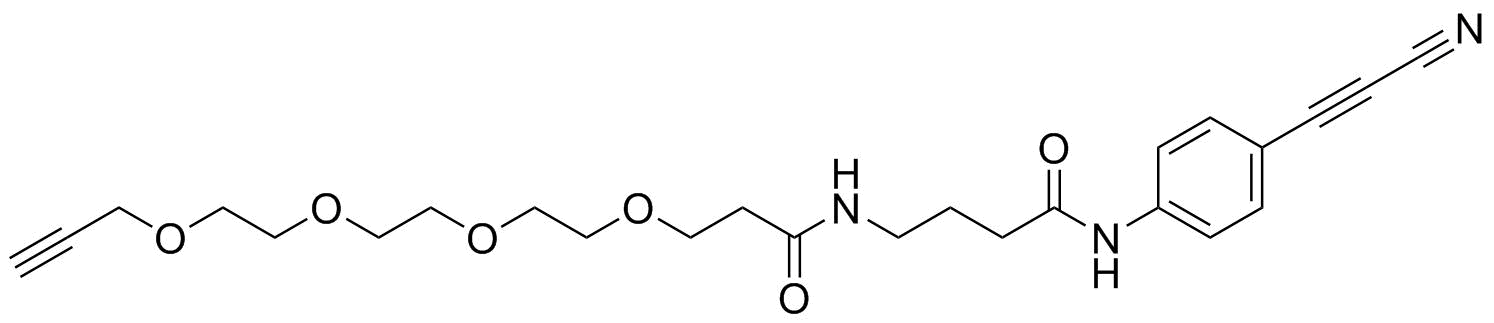
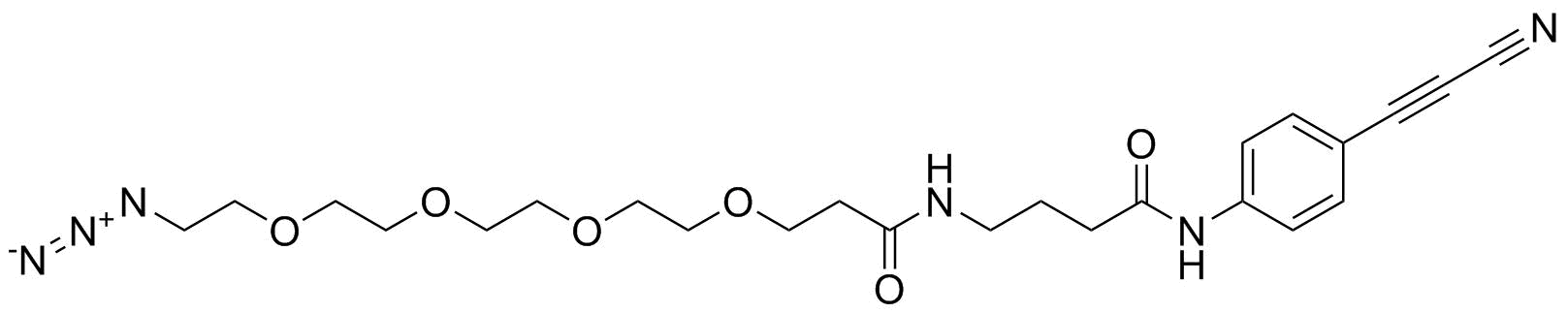
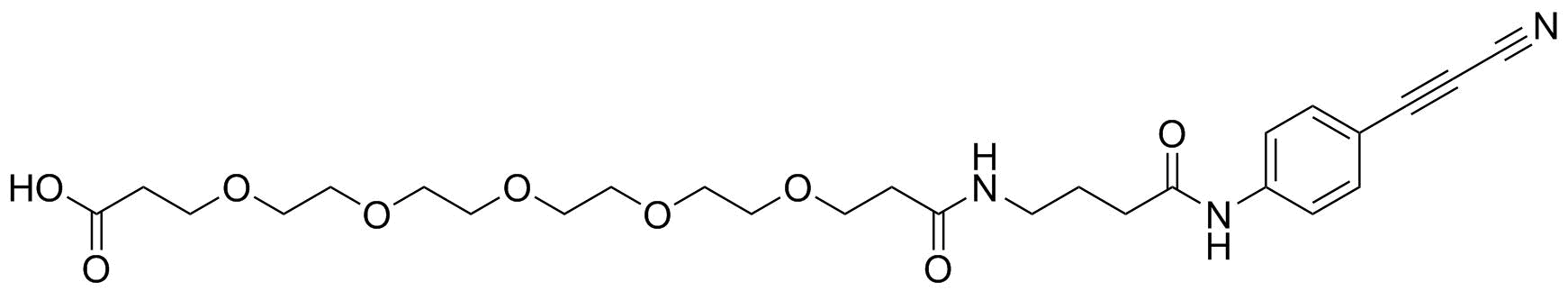
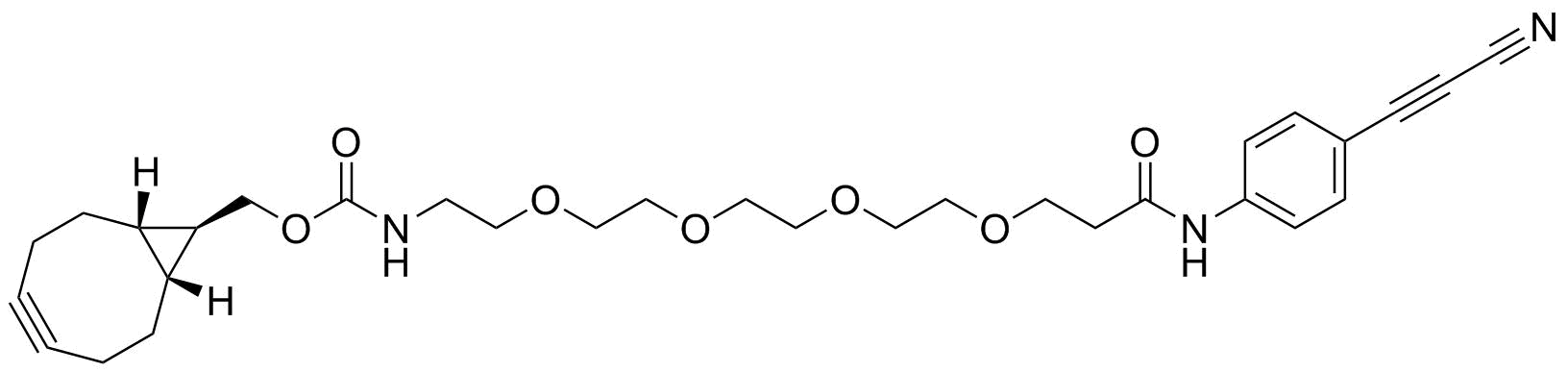
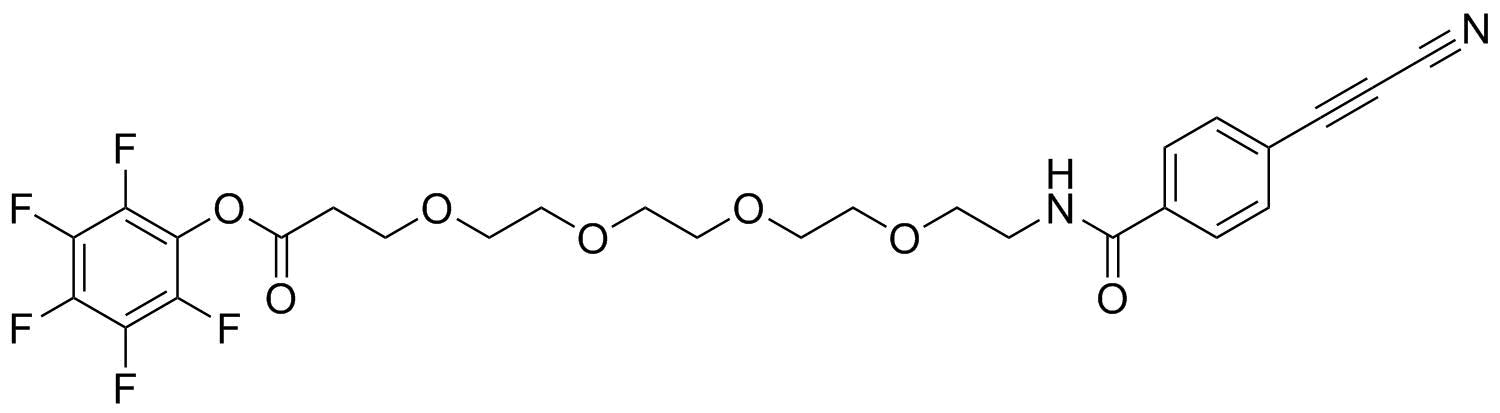
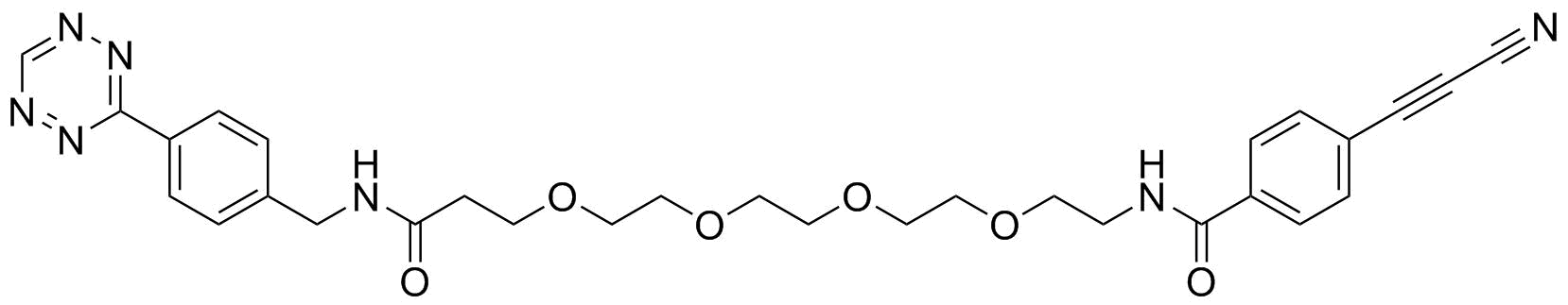
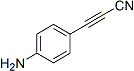APN linkers are designed to facilitate the conjugation of therapeutic agents, such as small molecules, peptides, or proteins, to a carrier molecule, such as an antibody or a nanoparticle. The resulting conjugate exhibits enhanced selectivity and efficacy in vivo, making APN linkers a promising tool for the development of targeted therapeutics.
APN linkers are characterized by the presence of two functional groups: an amino group (-NH2) and a carboxyl group (-COOH). These groups are reactive towards each other and can be coupled to form an amide bond (-CONH-) or an ester bond (-COO-). The choice of coupling chemistry depends on the nature of the therapeutic agent and the carrier molecule, as well as the desired stability and release profile of the resulting conjugate.
APN linkers have been employed in various drug delivery systems, such as antibody-drug conjugates (ADCs), which are designed to selectively target and kill cancer cells. In an ADC, the therapeutic agent is conjugated to an antibody via an APN linker, which ensures the drug is delivered specifically to cancer cells expressing the target antigen. The selectivity of the conjugate reduces toxicity to healthy cells, and the release of the drug is controlled, improving efficacy.